
Custom development
An agile scientific platform
Seqens provides active Pharmaceutical Ingredient (API) and drug substance development services. We support emerging and large pharmaceutical customers in human and animal health with their pharmaceutical and biotechnology applications for complex, niche and small molecule programs through two main R&D locations in the regions of Boston and Paris.

Early phase development
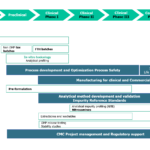
Seqens’ Development team operates state of the art R&D facilities to handle the most complex API development challenges. With a broad range of experience and knowledge of the latest technologies in organic synthesis, our chemists establish robust and economical routes for the synthesis of API.
- Route scouting
- Process development
- Process Safety
- Analytical development
- Preliminary ICH stability profile
- Non-GMP API supply for tox batches
- FTH and API clinical supply using cGMP kilo-lab suites
- IND document preparation & review (CMC or DMF preparation)
Special features
- Controlled substances
- Solid state services including polymorphism screening and salt selection
- Multicatalysis
- Biocatalysis
- Flow chemistry

Late phase development
Our R&D teams France & USA together with our Regulatory Affairs team in Ecully, France provide API cGMP services.
- Quality by design methodology
- Process and crystallization optimization studies
- Fate and purge studies
- Analytical method development and ICH validation
- Kilo-scale, pilot scale or multi-ton scale manufacturing to support phase III or launch
- non-GMP and GMP Raw material back integration
- ICH stability studies
Special features
- Cryogenic, very high pressure capabilities
- Flow Chemistry capabilities (up to 2 MT/year)
- Highly potent APIs down to 100 ng/m3
Our offer
Custom Synthesis & Polymerization
- Multicatalysis platform
- HTE (High Throughput Experiment) robotics platform
- Polymerization platform
- Microfluidics platform
- Hyphenated platforms with artificial intelligence
Selection, Isolation & Purification
- Solid forms screening
- Crystallization
- Isolation and Particle engineering
- Purification by chromatography
- High containment handling
Characterization & Analytical Science
- Solid state
- Pre-formulation
- Analytical impurities profiling
- Toxicological evaluation
Early Clinical formulation
- Preclinical Formulations/ Early-phase Formulations
- Bioavailability Improvement
- Controlled release profile
- Lipids & polymers synthesis as formulations excipients
- Early-phase forms delivery (capsules, sachets, granules…)
*Services consist of innovative (technical) early formulation activities on drug substances (APIs).
Any material supplies are not intended for clinical use.

SEQENS has extended its offer towards a comprehensive solution from Drug Substance to its early-phase formulation
– Development of phase-appropriate oral formulations
– Services to support your preclinical ADME studies
and first in human clinical trials (phases I and IIa)

Download our brochure
CMC project management
Our R&D teams in France and USA, are experienced in providing API CMC (Chemistry, Manufacturing and Controls) solutions, including IND-enabling, to meet your project needs.


R&D is at the heart of Seqens’ strategy. With 9 centers of excellence around the world, including more than 300 researchers, 130 of whom are PhDs, Seqens has unique capabilities to support its customers in the development of complex molecules across all markets.
